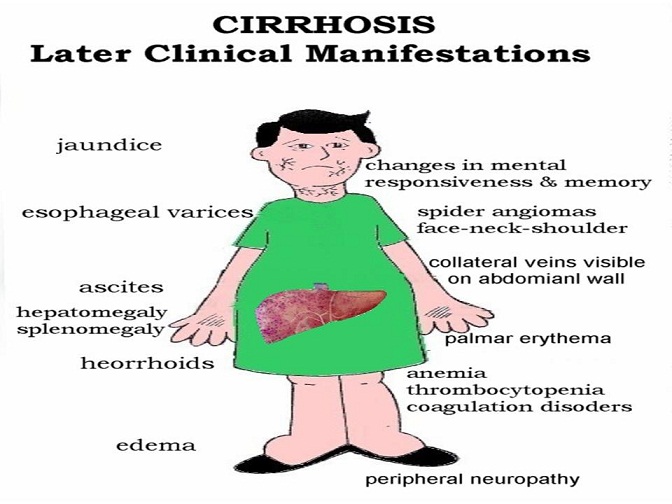
Complications of Cirrhosis: Ascites, Hepatic Encephalopathy, and Variceal Hemorrhage
Complications of Cirrhosis
Anuja Choure
William D. Carey
Ascites
Definition and Etiology
Ascites is defined as the accumulation of fluid in the peritoneal cavity. It is a common clinical finding, with various extraperitoneal and peritoneal causes (Box 1), but it most often results from liver cirrhosis. The development of ascites in a cirrhotic patient generally heralds deterioration in clinical status and portends a poor prognosis.
Box 1: Common Causes of Ascites
Extraperitoneal Causes
Budd-Chiari syndrome
Chylous ascites
Cirrhosis
Congestive heart failure
Hypoalbuminemia
Nephrotic syndrome
Malnutrition
Protein-losing enteropathy
Myxedema
Pancreatitis
Peritoneal Causes
Endometriosis
Infection
Tuberculosis
Bacterial
Fungal
Parasitic
Malignancy
Ovarian cancer
Pancreatic cancer
Other
Prevalence
Ascites is the most common major complication of cirrhosis and is an important landmark in the natural history of chronic liver disease. If observed for 10 years, approximately 60% of patients with cirrhosis develop ascites requiring therapy.
Pathophysiology
Cirrhotic ascites forms as the result of a particular sequence of events. Development of portal hypertension is the first abnormality to occur. As portal hypertension develops, vasodilators are locally released. These vasodilators affect the splanchnic arteries and thereby decrease the effective arterial blood flow and arterial pressures. The precise agent(s) responsible for vasodilation is a subject of wide debate; however, most the recent literature has focused on the likely role of nitric oxide.
Progressive vasodilation leads to the activation of vasoconstrictor and antinatriuretic mechanisms, both in an attempt to restore normal perfusion pressures. Mechanisms involved include the renin-angiotensin system, sympathetic nervous system, and antidiuretic hormone (vasopressin). The ultimate effect is sodium and water retention. In the late stages of cirrhosis, free water accumulation is more pronounced than the sodium retention and leads to a dilutional hyponatremia. This explains why cirrhotic patients with ascites demonstrate urinary sodium retention, increased total body sodium, and dilutional hyponatremia, a challenging concept for many physicians.
Signs and Symptoms
The symptoms of ascites vary from patient to patient and largely depend on the quantity of fluid. If trace ascites is present, the patient may be asymptomatic and fluid can be detected only on physical or radiologic examination. If a large amount of fluid is present, the patient might complain of abdominal fullness, early satiety, abdominal pain, or shortness of breath.
Physical examination findings are equally variable. The accuracy of detecting ascites depends on the amount of fluid present and the body habitus of the patient: ascites may be more technically difficult to diagnose in obese patients. If ascites is present, typical findings include generalized abdominal distention, flank fullness, and shifting dullness. If the physical examination is not definitive, abdominal ultrasonography can be used to confirm the presence or absence of ascites.
Two grading systems for ascites have been used in the literature (Table 1). An older system has graded ascites from 1+ to 4+, depending on the detectability of fluid on physical examination. More recently, a different grading system has been proposed, from grade 1 to grade 3. The validity of this grading system has yet to be established.
Table 1: Grading Systems for Ascites
Grade Severity Score
1 Minimal 1+
2 Moderate 2+
3 Severe 3+
4 Tense 4+
Diagnosis
If a noncirrhotic patient develops ascites, diagnostic paracentesis with ascites fluid analysis is an essential part of the medical evaluation. In a patient with well-established cirrhosis, the exact role of a diagnostic paracentesis is less clear. Our opinion is that for a highly functional outpatient with documented cirrhosis, the new development of ascites does not routinely require paracentesis. Cirrhotic patients should, however, undergo paracentesis in the case of unexplained fever, abdominal pain, or encephalopathy or if they are admitted to the hospital for any cause. It is common for hospitalized cirrhotic patients to have infected ascites fluid (spontaneous bacterial peritonitis, SBP) even if no symptoms are present. This is particularly true in the case of a significant gastrointestinal hemorrhage.
Complications from abdominal paracentesis are rare, occurring in less than 1% of cases. A low platelet count or elevated prothrombin time is not considered a contraindication, and prophylactic transfusion of platelets or plasma is almost never indicated. Insertion of the paracentesis needle is most commonly performed in the left or right lower quadrant, but it can also be performed safely in the midline. An abdominal ultrasound can guide the procedure if the fluid is difficult to localize or if initial attempts to obtain fluid are unsuccessful.
Valuable clinical information can often be obtained by gross examination of the ascites fluid (Table 2). Uncomplicated cirrhotic ascites is usually translucent and yellow. If the patient is deeply jaundiced, the fluid might appear brown. Turbidity or cloudiness of the ascites fluid suggests that infection is present and further diagnostic testing should be performed. Pink or bloody fluid is most often caused by mild trauma, with subcutaneous blood contaminating the sample. Bloody ascites is also associated with hepatocellular carcinoma or any malignancy-associated ascites. Milky-appearing fluid usually has an elevated triglyceride concentration. Such fluid, commonly referred to as chylous ascites, can be related to thoracic duct injury or obstruction or lymphoma, but it is often related primarily to cirrhosis.
Table 2: Gross Appearance of Ascites Fluid
Color Association
Translucent or yellow Normal/sterile
Brown Hyperbilirubinemia (most common)
Gallbladder or biliary perforation
Cloudy or turbid Infection
Pink or blood tinged Mild trauma at the site
Grossly bloody Malignancy
Abdominal trauma
Milky (“chylous”) Cirrhosis
Thoracic duct injury
Lymphoma
Box 2: Ascites Fluid Testing
Routine
Cell count with differential
Albumin
Total protein
Culture*
Sometimes Useful
Lactose dehydrogenase level
Glucose
Amylase
Triglyceride
Bilirubin
Cytology
Tuberculosis smear and culture
Rarely Helpful
Lactate
Gram stain
*If infection is suspected and/or corrected polymorphonuclear count is ≥250 cells/mm3.
Many ascites fluid tests are currently available, yet the optimal testing strategy has not been well established. Generally, if uncomplicated cirrhotic ascites is suspected, only a total protein and albumin concentration and a cell count with differential are determined (Box 2). Less than 10 mL of fluid is required to perform these basic tests. The albumin concentration is used to confirm the presence of portal hypertension by calculating the serum-to-ascites albumin gradient, or SAAG. The SAAG is determined by subtracting the ascites albumin value from a serum albumin value obtained on the same day:
AlbuminSerum – albuminascites = SAAG
The SAAG has been proved in prospective studies to categorize ascites better than any previous criteria. The presence of a gradient higher than 1.1 g/dL indicates that the patient has portal hypertension–related ascites with 97% accuracy. Portal hypertension is usually caused by liver cirrhosis or, less commonly, outflow obstruction from right-sided heart failure or Budd-Chiari syndrome. A SAAG value lower than 1.1 g/dL indicates that the patient does not have portal hypertension–related ascites, and another cause of the ascites should be sought. Determination of the SAAG does not need to be repeated after the initial measurement.
The cell count and differential are used to determine if the patient is likely to have SBP. Patients with an ascites polymorphonuclear (PMN) count greater than 250 cells/mm3 should receive empiric antibiotics, and additional fluid should be inoculated into blood culture bottles to be sent for culture. The PMN count is calculated by multiplying the white cells/mm3 by the percentage of neutrophils in the differential. In a bloody sample, which contains a high concentration of red blood cells, the PMN count must be corrected: 1 PMN is subtracted from the absolute PMN count for every 250 red cells/mm3 in the sample.
Based on clinical judgment, additional testing can be performed on ascites fluid including total protein, lactate dehydrogenase (LDH), glucose, amylase, triglyceride, bilirubin, cytology, or tuberculosis smear and culture. These tests are generally only useful when there is suspicion of a condition other than sterile cirrhotic ascites. Tests that are not routinely helpful include determination of pH, lactate levels, and Gram staining. Results of Gram staining are of particular low yield unless a large concentration of bacteria, such as in the case of a free gut perforation, is suspected.
Summary
Cirrhotic patients should undergo diagnostic paracentesis in cases of unexplained fever, abdominal pain, or encephalopathy or when admitted to the hospital for any cause.
Paracentesis is a safe procedure, with a low risk of serious complication.
The serum-to-ascites albumin gradient (SAAG) can be calculated to determine whether the patient has portal hypertension–related ascites.
The ascites fluid cell count and differential are used to determine whether the patient is likely to have spontaneous bacterial peritonitis (SBP).
Treatment
Successful treatment of cirrhotic ascites is defined as the minimization of intraperitoneal fluid without intravascular volume depletion. Despite a lack of data supporting decreased mortality, minimizing the amount of ascites fluid can decrease infection-related morbidity in the cirrhotic patient. Treatment of ascites can dramatically improve quality of life by decreasing abdominal discomfort or dyspnea, or both. General ascites management in all patients should include minimizing consumption of alcohol, nonsteroidal anti-inflammatory drugs (NSAIDs), and dietary sodium. The use of more-aggressive interventions largely depends on the severity of ascites and includes oral diuretics, therapeutic (or large-volume) paracentesis, transjugular intrahepatic portosystemic shunt (TIPS), and orthotopic liver transplantation (Fig. 1).
Low-Volume Ascites
All patients with cirrhotic ascites should be encouraged to minimize consumption of alcohol. Even if alcohol is not the cause of their liver disease, cessation can lead to decreased fluid and improved response to medical therapies. Patients with ascites should also minimize use of all NSAIDs; these agents inhibit the synthesis of renal prostaglandin and can lead to renal vasoconstriction, decreased diuretic response, and acute renal failure. Finally, ascites patients should be counseled to limit their sodium consumption to no more than 2 g/day. Because fluid passively follows sodium, a salt restriction without a fluid restriction is generally all that is required to decrease the amount of ascites. In patients with minimal fluid, the restriction of alcohol, NSAIDs, and salt may be all that is needed to control ascites formation adequately.
Moderate-Volume Ascites
Patients with moderate fluid overload who do not respond to more conservative measures should be considered for pharmacologic therapy. A rapid reduction of ascites is often accomplished simply with the addition of low-dose oral diuretics in the outpatient setting.
First-line diuretic therapy for cirrhotic ascites is the combined use of spironolactone (Aldactone) and furosemide (Lasix). Beginning dosages are 100 mg of spironolactone and 40 mg of furosemide by mouth daily. If weight loss and natriuresis are inadequate, both drugs can be simultaneously increased after 3 to 5 days to 200 mg of spironolactone and 80 mg of furosemide. To maintain normal electrolyte balance, the use of the 100 : 40 mg ratio of spironolactone to furosemide is generally recommended. Maximum accepted dosages are 400 and 160 mg/day of spironolactone and furosemide, respectively.
The response to diuretics should be carefully monitored on the basis of changes in body weight, laboratory tests, and clinical assessment. Patients on diuretics should be weighed daily; the rate of weight loss should not exceed 0.5 kg/day in the absence of edema and should not exceed 1 kg/day when edema is present. Serum potassium, blood urea nitrogen (BUN), and creatinine levels should be serially followed. In the event of marked hyponatremia, hyperkalemia or hypokalemia, renal insufficiency, dehydration, or encephalopathy, diuretics should be reduced or discontinued. Routine measurement of the urinary sodium level is not necessary, but it can be helpful to identify noncompliance with dietary sodium restriction. Patients excreting more than 78 mmol of sodium/day (88 mmol dietary intake − 10 mmol nonurinary excretion) detected on a 24-hour urinary collection should be losing fluid weight. If not, they are noncompliant with their diet and should be referred to a dietician. The spot urine sodium-to-potassium ratio might ultimately replace the cumbersome 24-hour collection: A random urine sodium concentration higher than the potassium concentration has been shown to correlate with a 24-hour sodium excretion higher than 78 mmol/day with approximately 90% accuracy. Because of the potentially severe complications associated with diuretic use, patients with ascites should be assessed by a health care provider at least once weekly until they are clinically stable.
Large-Volume Ascites
Large-volume ascites is defined as intraperitoneal fluid in an amount that significantly limits the activities of daily life. With additional fluid retention, the abdomen can become progressively distended and painful. This is commonly referred to as massive or tense ascites.
Therapeutic (or large-volume) paracentesis is a well-established therapy for large-volume ascites. However, the use of postprocedural colloid, usually albumin, continues to be a controversial issue. Studies have shown that patients who do not receive intravenous albumin after large-volume paracentesis develop significantly more changes in their serum electrolyte, creatinine, and renin levels. The clinical relevance of these findings, however, is not well established. In fact, no study to date has been able to demonstrate decreased morbidity or mortality in patients given no plasma expanders compared with patients given albumin after paracentesis. In view of the high cost of albumin and its uncertain clinical role, more studies certainly need to be conducted. Until these studies are carried out, current practice guidelines state that it is reasonable, although not mandatory, to give albumin for paracenteses greater than 5 L. Although no direct comparisons have been studied, 25% albumin at doses of 5 to 10 g/L of ascites removed is generally used.
To prevent the reaccumulation of ascites fluid, patients with large-volume ascites should be counseled about limiting consumption of alcohol, NSAIDs, and sodium. They should also be placed on an aggressive diuretic regimen. Diuretic-sensitive patients are generally treated with lifestyle modifications and medications, not serial paracentesis.
Refractory Ascites
Refractory ascites occurs in 5% to 10% of cirrhotic ascites patients and portends a poor prognosis. The definition of refractory ascites is (1) lack of response to high-dose diuretics (400 mg of spironolactone and 160 mg of furosemide/day) while remaining compliant with a low-sodium diet or (2) frequent ascites recurrence shortly after therapeutic paracentesis. Patients with recurrent side effects from diuretic therapy, including symptomatic hyponatremia, hyperkalemia or hypokalemia, renal insufficiency, or hepatic encephalopathy, are also considered to have refractory ascites. Treatment options include large-volume paracentesis with albumin infusion, placement of a TIPS, or liver transplantation. Surgical shunts (e.g., LeVeen or Denver shunt) have essentially been abandoned because controlled trials have shown poor long-term patency, excessive complications, and no survival advantage over medical therapy.
Frequent therapeutic paracentesis with or without albumin infusion is the most widely accepted treatment for patients with refractory ascites (see “Large-Volume Ascites” for controversy and dosing of albumin use). For those who have loculated fluid or are unwilling or unable to receive frequent paracentesis, TIPS placement can also be considered. In the appropriately selected patient, TIPS is highly effective for preventing ascites recurrence by decreasing the activity of sodium-retaining mechanisms and improving renal function. Ongoing studies will determine whether TIPS might also provide a survival benefit.
In the United States, TIPS is most commonly performed under conscious sedation by an interventional radiologist. The portal system is accessed through the jugular vein, and the operator inserts a self-expanding shunt between the portal (high-pressure) and hepatic (low-pressure) veins. The ultimate goal of the procedure is to lower portal pressures to less than 12 mm Hg, the level at which ascites begins to accumulate. Complications are relatively common and include hemorrhage (intrahepatic or intra-abdominal) and stent stenosis or thrombosis. Other important complications include hepatic encephalopathy and decompensation of liver or cardiac function. Therefore, TIPS is generally not recommended for patients with pre-existing encephalopathy, an ejection fraction lower than 55%, or a Child-Pugh Score higher than 12 (Table 3). Additional disadvantages of the procedure are high cost and lack of availability at some medical centers.
Table 3: Child-Pugh Classification*
Points
Clinical or Biochemical Parameter 1 2 3
Bilirubin (mg/dL) <2 2-3 >3
Albumin (g/dL) >3.5 2.8-3.5 <2.8
Ascites Absent Moderate Tense
Encephalopathy Absent Moderate (I or II) Severe (III or IV)
Prothrombin time
Seconds prolonged or <4 4-6 >6
International normalized ratio (INR) <1.7 1.7-2.3 >2.3
*Child-Pugh score: A = 5-6, B = 7-9, C = 10-15.
Liver transplantation is the ultimate treatment for cirrhosis and cirrhotic ascites. Appropriate timing for referral is debated, but should be considered when a cirrhotic patient first presents with a complication from cirrhosis, such as ascites. Because refractory ascites portends a particularly poor prognosis, immediate referral to an experienced liver transplantation center is recommended.
The 2-year survival rate for a patient with cirrhotic ascites is approximately 50%. Once a patient becomes refractory to routine medical therapy, 50% die within 6 months and 75% within 1 year. Because liver transplantation is associated with 2-year survival rates of almost 85%, it should be considered as an important treatment option in all appropriate patients.
Many with ascites will develop infection, most often without a known precipitating factor (such as diverticulitis, bowel perforation, etc). This is referred to as spontaneous bacterial peritonitis (SBP). SBP should be suspected whenever there is clinical deterioration in a cirrhotic with ascites. Diagnosis rests on ascitic fluid cell count (more than 250 polymorphonuclear cells/mm3) or a positive ascitic fluid culture. Treatment should be undertaken whenever SBP is suspected. Most often intravenous therapy with a third undertaken whenever SBP is suspected. Most often intravenous therapy with a third generation cephalosporin (e.g., cefotaxime 2 gm every 8 hours) is used. Quinolones may also be effective, including oral agents such as ofloxacin (400 mg twice per day). Intravenous albumin (1.5 gm/kg body weight on day zero and 1.0 gm/kg on day 3 has been shown to improve survival in SBP, particularly in those with renal insufficiency and should be used if the creatinine or BUN are elevated. Antibiotic treatment should be continued for 5 days. The presence of bacteremia does not influence treatment duration. Longer therapy may be warranted in individual cases. Those who survive SBP are at risk for a second bout. Prophylactic antibiotics are recommended, e.g., norfloxacin 400 mg daily, or trimethoprim/sulfamethoxazole (160/800). A clinical practice guideline updated in 2009 suggests a role for SBP prophylaxis even in those who have never had SBP. Primary SBP prophylaxis should be considered in patients with ascites containing <1.5 gm/dl and one or more of the following: serum creatinine >1.2 mg/dl; BUN >25 mg/dl; serum sodium concentration <130 mEq/L, Childs Pugh score >9 together with a bilirubin >3 mg/dl.
Hepatorenal Syndrome
A feared complication of advanced liver disease is hepatorenal syndrome (HRS). In its most virulent form (type I) there is an inexorable worsening of renal function reflecting in rising creatinine and BUN, resulting in death. This syndrome almost always occurs in the setting of ascites. A diagnosis is established when other causes of acute renal insufficiency are excluded, especially hypovolemia due either to diuretic use, infection, or bleeding. HRS is established in a patient with a creatinine of >1.5 mg/dl that does not improve with withdrawal of diuretics, volume expansion with albumin, absence of obstructive or parenchymal renal disease (indicated by ultrasonography, proteinuria), and absence of recent use of nephrotoxic drugs or administration of IV contrast material. Treatment of HRS is frustrating and often unsuccessful. Studies suggest a possible role for use of intravenous albumin (e.g., 25 grams daily) together with octreotide (200 ug sc TID) and midodrine (5 mg TID, titrating to a maximum of 12.5 mg TID). Referral for liver transplantation services should be considered for appropriate patients. A less severe form of HRS (type II) is recognized. In this variant, non-progressive renal impairment without another etiology than cirrhosis is seen.
Hepatic encephalopathy
Definition and Etiology
Hepatic encephalopathy (HE) is defined as mental or neuromotor dysfunction in a patient with acute or chronic liver disease. Several forms of HE have been described (Box 3). The acute form of HE is often associated with fulminant hepatic failure and can rapidly progress to seizures, coma, and death. In patients with cirrhosis, acute encephalopathy is most commonly associated with a precipitating factor, such as electrolyte disturbance, medications, gastrointestinal hemorrhage, or infection. Recurrent HE can occur with or without a precipitating factor and is usually easily reversible. Persistent HE is rare and is defined as the persistence of neuropsychiatric symptoms despite aggressive medical and dietary therapy. The most common form of HE is not always clinically apparent: A patient with subclinical HE has only mild cognitive deficits or subtle personality changes. Recent evidence demonstrates individuals with even miximal hepatic encephalopathy have impacted driving skills and a high rate of motor vehicle accidents. Specific neuropsychological or neurophysiologic testing may be required to secure the diagnosis in these cases.
Box 3: Precipitating Factors in Hepatic Encephalopathy
Anemia
Azotemia, uremia
Constipation
Dehydration
Excessive dietary protein
Gastrointestinal bleeding
Hepatoma
Hypokalemia, metabolic alkalosis
Hypoglycemia
Hypothyroidism
Hypoxia
Infection (e.g., urinary tract, ascites)
Medications (e.g., narcotics, sedatives)
Vascular occlusion
Prevalence
HE may be clinically apparent in as many as one third of cirrhotic patients and, if rigorously tested, up to two thirds have some degree of mild or subclinical HE.
Pathophysiology
The exact etiology of how HE develops is largely unknown. The premise of most pathophysiologic theories involves the accumulation of ammonia in the central nervous system, producing alterations of neurotransmission that affect consciousness and behavior. These ammonia toxicity theories have been supported by studies demonstrating increased ammonia levels in patients with both fulminant hepatic failure and chronic liver disease. The lack of strong correlation between serum ammonia levels and stage or degree of encephalopathy has been used in the argument that hyperammonemia might not be the sole factor in HE pathogenesis.
Most ammonia is produced in the intestine by colonic breakdown of nitrogenous compounds and enterocytic catabolism of amino acids. Other sources of ammonia are the kidneys and skeletal muscle. Normally, ammonia is metabolized in the liver and promptly excreted through the kidneys or colon. Formation of glutamine from glutamate, by glutamine synthetase, in the liver and brain is another means of detoxifying ammonia. Impaired liver function, shunting of blood around the liver, and increased muscle wasting all lead to increased serum ammonia levels in cirrhotic patients.
Ammonia interferes with brain function at many sites. Ammonia crosses the blood-brain barrier and directly depresses the central nervous system. There is also evidence that hyperammonemia might facilitate the brain’s uptake of tryptophan, a substance with neuroactive metabolites such as serotonin. Excess ammonia can reduce brain adenosine triphosphate (ATP) levels, resulting in impaired cerebral energy. Lastly, the metabolism of ammonia to glutamine in the brain increases the intracellular osmolarity of astrocytes, inducing astrocyte swelling and vasodilation. Increased astrocyte hydration without overt increased intracranial pressure is currently considered a major factor in the development of HE in patients with chronic liver disease.
Toxins other than ammonia have also been implicated in the pathogenesis of HE. Excesses of neurotoxic short-chain fatty acids and mercaptans have received attention in the past. Patients with cirrhosis have also been shown to have decreased branched-chain amino acid (BCAA) to aromatic amino acid (AAA) ratios. It has been postulated that the increased AAA level in the cirrhotic patient’s brain might competitively inhibit normal neurotransmitters. Cirrhotic patients with HE have been shown to have greatly increased serum manganese levels. Manganese can deposit directly in the basal ganglia and induce extrapyramidal symptomatology. Manganese can also act synergistically with ammonia to activate peripheral-type benzodiazepine receptors and the gamma-aminobutyric acid (GABA)-ergic neuroinhibitory system.
Signs and Symptoms
In patients with progressive HE, there is a gradual decrease in level of consciousness, intellectual capacity, and logical behavior, along with the development of specific neurologic deficits. Two staging systems have been described. Numerous studies have used the West Haven criteria of altered mental status in patients with HE (Table 4). Although the Glasgow coma scale has not been rigorously evaluated in this specific patient population, its widespread use in various other disorders of brain function makes it applicable for patients with acute or chronic liver disease (Table 5).
Table 4: Clinical Characteristics of Various Forms of Hepatic Encephalopathy
Form Precipitating Factors Clinical Course Reversibility
Acute + Short* ±*
Recurrent ± Short +
Persistent − Continuous −
Subclinical − Insidious −
*May be fatal or irreversible as in fulminant hepatic failure.
Table 5: West Haven Criteria of Altered Mental Status in Hepatic Encephalopathy
Stage Consciousness Intellect and Behavior Neurologic Findings
0 Normal Normal Normal examination
Impaired psychomotor testing
1 Mild lack of awareness Shortened attention span
Impaired addition or subtraction Mild asterixis or tremor
2 Lethargic Disoriented
Inappropriate behavior Obvious asterixis
Slurred speech
3 Somnolent but arousable Gross disorientation
Bizarre behavior Muscular rigidity and clonus
Hyperreflexia
4 Coma Coma Decerebrate posturing
Diagnosis
HE is a diagnosis of exclusion. Similar neuropsychiatric symptoms are seen in various metabolic disorders, toxic ingestions, or intracranial processes (Table 6). In certain patients, brain imaging or electroencephalography (EEG) may be indicated to exclude an intracranial abnormality. Lumbar puncture with cerebrospinal fluid analysis may also be required for patients with unexplained fever, leukocytosis, or symptoms suggesting meningeal irritation. Knowledge of the existence of acute or chronic liver disease, history of HE, or both is often helpful in heightening clinical suspicion and securing the diagnosis.
Table 6: Glasgow Scale of Level of Consciousness
Best Motor Response (M) Score Best Verbal Response (V) Score Eyes Open (E) Score
Obeys verbal orders 6
Localizes painful stimuli 5 Oriented and conversant 5
Withdraws from painful stimuli 4 Disoriented and conversant 4 Spontaneously 4
Flexion in response to pain 3 Inappropriate words 3 To command 3
Extension in response to pain 2 Inappropriate sounds 2 To pain 2
No response 1 No response 1 No response 1
M + V + E = 3 to 15. Severe encephalopathy is defined as a score ≤12.
The most commonly studied laboratory test in HE is the venous ammonia level. Because of inconsistent elevation and lack of correlation with the stage of encephalopathy, determination of ammonia levels is not considered to be a good screening tool. Measurement of serum ammonia can be helpful, for example, when the level is elevated and there is doubt regarding the presence of significant liver disease. The arterial ammonia concentration provides a more accurate assessment of the amount of ammonia at the blood-brain barrier, but it is also of limited clinical use.
Because of implications in diagnosis and treatment, a search for a precipitating factor should be sought in all cirrhotic patients hospitalized for HE (Box 4). Serum determination of the complete blood count, electrolyte levels, and renal function is indicated in almost all cases. Recent use of psychoactive medications, such as narcotics or sedatives, should also be investigated. In a confused patient who cannot give a reliable history, the examination of stool or placement of a nasogastric tube may help in detecting gastrointestinal bleeding. Because infection is a common precipitating factor in HE, the culture of body fluids (urine, blood and ascites, if present) should be routinely performed. Lastly, the consumption of excessive dietary protein and constipation can precipitate HE. This is believed to be the result of an increased nitrogen load in the gastrointestinal tract. Rare precipitants, such as hepatoma or vascular occlusion, need only be investigated if no other factors are believed to be contributing or with clinical suspicion.
Box 4: Differential Diagnostic Considerations in Hepatic Encephalopathy
Metabolic
Hypo- or hyperglycemia
Hypo- or hypercalcemia
Hypokalemia
Hypoxia
Uremia
Toxic
Alcohol intoxication
Alcohol withdrawal
Carbon monoxide narcosis
Illicit drugs
Medications
Central Nervous System
Bleed or infarction
Abscess, meningitis
Encephalitis
Trauma
Tumor
Summary
HE is a diagnosis of exclusion.
Determination of a serum ammonia level can be helpful but is not a good screening tool.
All cirrhotic patients hospitalized for HE should be evaluated for a precipitating factor.
Treatment
The main objectives in the treatment of HE are fourfold: provide supportive care, correct any precipitating factors, reduce the nitrogen load in the gastrointestinal tract, and assess the need for long-term therapy. Each of these objectives is discussed separately here.
Standard supportive care is required for all hospitalized patients with HE. Patient safety and frequent monitoring of mental status are crucial. This can require additional personnel and, in the case of comatose patients, admission to the intensive care unit, endotracheal intubation, or both. Patients with HE should also avoid prolonged periods of fasting. Although the restriction of dietary protein at the time of acute HE can be part of therapy, protracted nitrogen restriction can lead to malnutrition. Appropriate enteral nutrition, by mouth or nasogastric feeding tube, should be administered as soon as feasible.
A methodical search to identify and treat any precipitating factors is crucial in reversing the signs and symptoms of HE. (See earlier, “Diagnosis,” for a more detailed discussion.)
Because the toxins believed to be responsible for HE arise in the gastrointestinal tract, removal of the nitrogenous load is the mainstay of therapy. Various pharmacologic agents may be used, but the nondigestable disaccharide known as lactulose is currently the first-line therapy. After consumption, lactulose passes through the small bowel completely undigested. Once in the colon, lactulose is metabolized by colonic bacteria and the pH is lowered. This acidification of the bowel is believed to underlie the cathartic effect; ammonia can then pass from the blood stream into the colonic lumen to be excreted. As a result, peripheral ammonia levels are reduced.
For acute encephalopathy, lactulose can be administered either orally (by mouth or through a nasogastric tube) or via retention enemas (Table 7). The usual oral dose is 45 mL followed by dosing every hour or two until evacuation occurs. At that point, dosing is adjusted to attain two or three soft bowel movements daily. This usually requires 15 to 45 mL every 6 to 12 hours. Lactulose by enema is administered as 300 mL in 1 L of water and should be retained for 1 hour. Because of the difficulty of administration, lactulose by enema is not generally used for chronic therapy. Common side effects include flatulence, bloating, and diarrhea.
Table 7: Commonly Used Medications for Hepatic Encephalopathy
Drug Dose (Acute) Dose (Chronic) Side Effects
Lactulose
Oral 45 mL PO q1-2hr 15-45 mL PO q6-12hr Diarrhea, flatulence, cramps
Enema 300 mL in 1 L of water q4-6hr Not used chronically Diarrhea, flatulence, cramps
Antibiotics
Metronidazole 250 mg PO q8-12hr 250 mg PO q8-12hr Peripheral neuropathy
Rifaximin 400 mg PO q8hr 400 mg PO q8hr Rare flatulence, constipation
Neomycin 1000 mg PO q4-8hr 500 mg PO q6-12hr Rare nephro- and ototoxicity
For patients who do not tolerate lactulose or have continued symptoms, antibiotics are a second-line alternative for therapy. Antibiotics are believed to reverse HE by its alteration of colonic bacteria. Metronidazole (Flagyl) and neomycin are most commonly used. Metronidazole is generally administered at 250 mg every 8 to 12 hours. Dose and duration of metronidazole should be minimized as much as possible to avoid peripheral neuropathy, a side effect associated with its long-term use. Neomycin is as effective as metronidazole and is administered at 3 to 6 g/day in acute encephalopathy and 1 to 2 g/day when used chronically. Neomycin use should also be limited, if possible, because long-term use can lead to rare ototoxicity, renal failure, or both. If administered chronically to control HE symptoms (with or without lactulose), periodic renal and annual auditory monitoring should be performed.
Rifaximin (Xifaxon®) is a nonabsorbable derivative of rifampin that has been used in Europe for a wide variety of gastrointestinal problems, including HE, for many years. In 2005, the drug received approval from the U.S. Food and Drug Administration (FDA) for HE therapy in the United States. Previous studies have demonstrated that rifaximin at doses of 400 mg every 8 hours is as effective as lactulose and neomycin at improving HE symptoms. Rifaximin has few side effects, making it an appealing alternative to other antibiotics, particularly when prolonged therapy is required.
Several other HE therapies have been investigated, but they are not routinely used. These include zinc, ornithine aspartate, flumazenil, and bromocriptine. In experimental models, zinc supplementation has been shown to decrease ammonia levels. Human studies, however, are conflicting, and zinc is currently only recommended in patients with known zinc deficiency. Some studies using ornithine aspartate have been encouraging, but the drug is not currently available in the United States. Flumazenil may also be helpful, but it is currently only indicated for patients with acute HE and suspected benzodiazepine intake. No oral or long-acting preparations are currently available, which would make flumazenil cumbersome to use long term. Improvements of extrapyramidal symptoms have also been reported when bromocriptine was added to more conventional therapies. Bromocriptine, at 30 mg orally twice daily, can be considered in patients who are refractory to other therapies.
Occasionally, a portosystemic shunt (spontaneous, surgical, or from placement of a TIPS) is believed to be the primary cause of recurrent or chronic HE. In these rare cases, the shunt can be occluded. This is generally accomplished by placement of occlusive coils by an interventional radiologist. This advanced procedure should only be undertaken at experienced medical centers and after all other measures have failed.
Before discharge from the hospital, all cirrhotic patients with HE should be assessed for the need for long-term therapy. Patients should be counseled on avoiding precipitating factors such as constipation and psychoactive medications. Compliance with chronic medications, including lactulose, antibiotics, or both, should be emphasized. They should also be counseled about their higher risk of motor vehicle accidents and advised to curtail driving of appropriate. Appropriate candidates should be referred to a liver transplantation center after the first episode of overt encephalopathy. The ultimate therapy for cirrhosis and HE is orthotopic liver transplantation.
Some forms are reversible, but the development of overt HE overall carries a poor prognosis. Recovery and recurrence rates are variable but, without liver transplantation, the 1-year survival is only 40%. Both acute and chronic HE, once advanced to stage 4 (coma), is associated with an 80% overall mortality rate.
Summary
The main objectives in the treatment of HE are to provide supportive care, correct any precipitating factors, reduce the nitrogen load in the gastrointestinal tract, and assess the need for long-term therapy.
Lactulose is generally considered first-line therapy for acute and chronic HE.
Antibiotics are second-line therapy for HE, but they can have many potential side effects and complications.
Rifaximin was recently FDA approved for use in HE and appears to have fewer side effects than other antibiotics.
Development of overt HE is a poor prognostic indicator.
Variceal Hemorrhage
Definition and Etiology
Varices are dilated submucosal veins, most commonly detected in the distal esophagus or proximal stomach, but they can occur anywhere in the gastrointestinal tract. Varices are associated with portal hypertension of any cause, including cirrhosis. Any cirrhotic patient with varices is placed at risk for variceal hemorrhage, the most lethal complication of cirrhosis. Despite advances in therapy over the last decade, variceal hemorrhage is associated with a mortality of at least 20% at 6 weeks.
Prevalence
Esophageal varices are present in approximately 50% of patients with cirrhosis. Gastric varices are less prevalent, occurring in 5% to 33% of these patients. Variceal hemorrhage occurs at a yearly rate of 5% to 15%. Clinical and endoscopic predictors of a first variceal hemorrhage include advanced Child-Pugh score (see Table 3), continued alcohol use, large varices, and presence of red wale markings.
Pathophysiology
In cirrhosis, portal pressures initially increase as a consequence of resistance to blood flow within the liver. This resistance is due mainly to fibrous tissue and regenerative nodules in the hepatic parenchyma. In addition to this structural resistance, there is intrahepatic vasoconstriction, as well. This is believed to be due to decreased production of endogenous nitric oxide.
Varices are the portosystemic collaterals that form after pre-existing vascular channels are dilated by portal hypertension. Dilation generally is clinically significant once the wedged hepatic venous pressure (WHVP) gradient is elevated above 12 mm Hg (normal, <5 mm Hg). The WHVP is defined as the gradient between the wedged hepatic venous pressure and the free hepatic venous pressure measured by a catheter threaded down through the jugular vein. It is important to remember that varices do not invariably form once the WHVP is above 12 mm Hg, so this pressure gradient is generally considered necessary but not sufficient.
Signs and Symptoms
Nonbleeding varices are generally asymptomatic. Once varices are bleeding, patients classically present with symptoms of an upper gastrointestinal hemorrhage such at hematemesis, passage of black or bloody stools, lightheadedness, or decreased urination. Associated signs of variceal hemorrhage include decompensated liver function manifested as jaundice, hepatic encephalopathy, worsened or new-onset ascites. Physical examination will likely reveal hypotension or shock (in severe cases), pallor and stigmata of chronic liver disease such as spider angiomatas, palmar erythema, gynecomastia, or splenomegaly. A rectal examination should be performed on all patients without obvious bleeding. A black tarry stool on the gloved finger suggests an upper gastrointestinal source, and further workup needs to be pursued. Hemoccult testing is not necessary because clinically significant bleeding should be apparent with visual inspection of the stool alone.
Diagnosis
Endoscopic surveillance for esophageal varices in cirrhosis.
The gold standard for the diagnosis of varices is esophagogastroduodenoscopy (EGD). It is generally recommended that patients with cirrhosis undergo elective endoscopic screening for varices at the time of diagnosis and periodically thereafter if no or small varices are detected (Fig. 2). If screening EGD reveals appreciable esophageal varices, a size classification should be assigned. Different size classification systems have been used over the years; however, a recent consensus meeting proposed that varices be categorized in only two grades, small and large. An appropriate cut-off was determined to be 5mm; that is, small varices are those less than 5 mm and large varices are those greater than 5 mm.
Another procedure that is currently being studied for screening for varices is esophageal capsule endoscopy. Pilot studies suggest it is safe and well tolerated (and does not require sedation), although its sensitivity and cost effectiveness still needs to be established.
EGD is also the main method for diagnosing variceal hemorrhage. The diagnosis is secured when endoscopic evaluation reveals active bleeding from a varix or clinical evidence of a recent bleed, such as an overlying clot. Esophageal or gastric varices should be indicted as the most likely source of hemorrhage when varices are present and no other potential source of bleeding is found.
Treatment
Practice guidelines have recently been updated regarding the prevention and management of gastroesophageal varices and variceal hemorrhage in cirrhosis. Treatment of varices is best considered in three distinct phases: prevention of the first variceal hemorrhage (primary prophylaxis), control of acute hemorrhage, an prevention of a second hemorrhage in a patient who has already bled (secondary prophylaxis).
Primary Prophylaxis
If a patient has small varices that have never bled and has no risk factors for a first variceal hemorrhage (high Child-Pugh score, continued alcohol use, presence of red wale markings), prophylactic strategies can be considered, although the long-term benefit has never been established. In our practice, primary prophylaxis for bleeding has often been reserved for high-risk patients who have small varices and for all patients with large varices.
The primary pharmacologic strategy for preventing variceal hemorrhage is use of nonselective beta blockers, particularly propranolol and nadolol. These medications reduce portal pressures both by decreasing cardiac output and by producing splanchnic vasoconstriction. Several studies have shown that nonselective beta blockers decrease the risk for first variceal hemorrhage by 40% to 50% when compared to patients taking placebo. A meta-analysis has also showed a statistically significant decrease in overall mortality. Selective beta-blockers, such as atenolol and metoprolol, are less effective and are not currently recommended for primary prophylaxis. Likewise, use of isosorbide mononitrate (alone or with nonselective beta blockers) is not currently recommended.
Propranolol is usually started at a dose of 20 mg twice daily and nadolol at a dose of 40 mg daily. The goal of therapy is to reduce the WHVP by at least 20% or to a gradient of less than 12 mm Hg. However, because WHVP measurement is not widely available, most clinicians aim to titrate the dose of beta blockers to achieve a resting heart rate of 55 beats/min or a reduction of heart rate by 25% from baseline. Unfortunately, beta blockers have some significant side effects, so often the dose is simply adjusted to a maximally tolerated dose. The most common side effects reported are lightheadedness, fatigue, shortness of breath, and impotence in men. Relative contraindications to the use of beta blockers include reactive airways disease, insulin-dependent diabetes (with episodes of hypoglycemia), and peripheral vascular disease.
Patients who meet criteria for primary prophylaxis but who cannot tolerate or have contraindications to beta blocker therapy should be considered for prophylactic endoscopic variceal ligation (EVL). Although studies have been conflicting, a recent consensus panel of experts concluded that both nonselective beta blockers and EVL are effective in preventing first variceal hemorrhage. The decision on whether to treat pharmacologically or via EVL should be based on patient characteristics and preferences, local resources, and expertise.
Acute Variceal Hemorrhage
Cirrhotic patients with suspected acute variceal hemorrhage should be admitted directly to an intensive care unit setting for frequent monitoring and aggressive management (Fig. 3). While still in the emergency department, initial resuscitation can begin by securing large-bore IVs and sending bloodwork to the lab, including a type and crossmatch for blood products. Volume resuscitation should be undertaken promptly but with caution because vigorous resuscitation can actually increase portal pressures to levels higher than baseline, thereby prompting rebleeding. In our practice we usually start the resuscitation with normal saline and switch to blood or albumin (or both), once available, with the goal to maintain hemodynamic stability. Transfusion of fresh frozen plasma and platelets can be considered in patients with a severe coagulopathy or thrombocytopenia. Low threshold should be taken to intubate the patient for airway protection, particularly if the patient is in shock or encephalopathic, because aspiration of blood often occurs.
Mangement of acute variceal hemorrhage.TIPS, transjugular intrahepatic portosystemic shunt.
Antibiotics are routinely administered in cirrhotic patients who are admitted to the hospital with variceal hemorrhage. Several randomized clinical trials were able to show that antibiotics not only decreased the rate of bacterial infection in these patients but also decreased the incidence of early rebleeding and increased overall survival. The optimal antibiotic and duration is unclear, because benefit was detected from many different regimines. In general, oral norfloxacin at doses of 400 mg twice daily for 7 days or IV ciprofloxacin (in patients in whom oral administration is not possible) is the recommended antibiotic. In patients with advanced cirrhosis or at hospitals with a high incidence of quinolone resistance, ceftriaxone at a dose of 1 g IV daily may be preferable.
Pharmacologic therapy to decrease portal pressures is critically important and should be considered the first-line treatment for acute variceal hemorrhage. It should be initiated as soon as the diagnosis of variceal hemorrhage is suspected and before EGD. The most common pharmacologic agent used in the United States for this purpose is octreotide, a somatostatin analogue that causes splanchnic vasoconstriction. The advantage of octreotide is that it can stop variceal hemorrhage in up to 80% of patients and is nearly devoid of side effects. It has been most widely used as an initial IV bolus of 50 µg followed by 50 µg/hour. This agent should be administered ideally for 5 days, even after bleeding is controlled. Vasopressin (most often used with nitroglycerin) is the most potent splanchnic vasoconstrictor, but it is rarely used for control of variceal hemorrhage due to its multiple vascular side effects including myocardial and mesenteric ischemia and infarction. Terlipressin is a vasopressin analogue that has significantly fewer side effects. It is effective in controlling variceal hemorrhage and reducing mortality. It is administered at an initial dose of 2 mg IV every 4 hours and then titrated down to 1 mg every 4 hours once bleeding is controlled. Terlipressin is currently used extensively in other parts of the world but is not widely available in the United States.
Even though pharmacologic therapy can be effective at controlling suspected variceal hemorrhage, EGD should be performed as soon as possible to confirm the diagnosis and implement endoscopic therapy. Endoscopic therapy is highly effective and can control variceal bleeding in 80% to 90% of patients. Sclerotherapy, widely used in the past, is now nearly obsolete because of improvement in EVL devices and risk of complication. Indeed, recent consensus determined EVL to be the preferred form of endoscopic therapy for acute esophageal variceal bleeding, although sclerotherapy is still recommended in patients in whom EVL is not technically feasible. Gastric varices, which are often not amenable to either EVL or sclerotherapy, may be more difficult to treat. N-butyl-2-cyanoacrylate glue injected directly into the varix has been shown to be effective for control of bleeding gastric varices.
Despite endoscopic and pharmacologic therapies, variceal bleeding cannot be controlled or recurs in up to 20% of patients. It is at this time that portal decompressive therapy, either shunt surgery or TIPS, can be considered. As TIPS has become more widely available, this is becoming the preferred decompressive procedure. However, performance of either TIPS or shunt surgery largely depends on local expertise. Because TIPS and surgery are both invasive procedure with a high risk of complication, they are reserved for patients who fail pharmacologic therapy and repeated attempts at endoscopic therapy.
Balloon tamponade applies direct pressure to the ruptured varix and can be highly effective for immediate control of variceal hemorrhage. Unfortunately, recurrent bleeding is common after the balloon is decompressed, and balloon tamponade is associated with potentially fatal complications such necrosis or perforation of the esophagus. Therefore, tamponade should be used only as a rescue procedure and a bridge to more definitive therapy, such as TIPS, in the cases of uncontrolled bleeding.
Secondary Prophylaxis
Patients who survive an episode of acute variceal hemorrhage are at high risk of rebleeding and death. If bleeding is left untreated, the rebleeding rate is nearly 60% within 1 to 2 years, with a mortality rate of 33%. Several studies have demonstrated that combination endoscopic plus pharmacologic therapy is the most effective means of preventing secondary bleeding episodes.
In terms of endoscopic therapies, EVL is the method of choice for secondary prophylaxis. After inital control of the bleeding, EVL should be repeated at 1- to 2-week intervals until varices are completely obliterated. This usually requires 2 to 4 sessions. Once the varices are obliterated, EGD is repeated every 3 to 6-months to evaluate the need for repeat EVL. Complications of EVL include chest pain, dysphagia and ulcers that form at the site of the band ligation, which universally form and can cause significant bleeding. Although not definitively proven to be effective, proton pump inhibition is sometimes used in an attempt to decrease the bleeding risk from these band ulcer sites for 2 weeks after an EVL procedure.
Optimal pharmacologic therapy for secondary prophylaxis appears to be a combination of a nonselective beta blocker and a nitrate. However, this combination has significantly greater side effects compared to beta blockers alone and is overall poorly tolerated. In our clinical practice, most patients end up taking beta blockers alone. Clinical opinion is divided on the need to continue pharmacologic therapy once varices are completely obliterated, but current guidelines suggest that pharmacologic therapy should be continued at the highest tolerated dose indefinitely. TIPS or shunt surgery can be considered in patients who experience recurrent bleeding despite combination pharmacologic and endoscopic therapy.
Most variceal haemorrhages can be controlled with these measures. However, because acute variceal bleeding often precipitates a clinical deterioration and worsening of liver synthetic function, patients who are otherwise transplant candidates should be referred to a liver transplantation center for a liver transplant evaluation after recovery.
Summary
Cirrhosis sets the stage for risk of GI bleeding once the WHVP rises above 12 mm Hg.
Primary prophylaxis with either nonselective beta blockers or EVL is warranted in cirrhotic patients with small varices and high-risk features and in all patients with large varices.
Treatment of acute variceal hemorrhage involves careful volume resuscitation, administration of antibiotics and drugs to reduce portal hypertension and prompt endoscopy therapy.
Combination of pharmacologic and endoscopy therapy is employed for secondary prevention of variceal hemorrhage.
TIPS and surgical shunts can be considered in patients in whom recurrent variceal hemorrhage occurs despite maximal pharmacologic and endoscopic therapies.
Suggested Readings
Ascites
Ginès P, Cárdenas A, Arroyo V, Rodés J. Management of cirrhosis and ascites. N Engl J Med. 2004, 350: 1646-1654.
Ginès P, Quintero E, Arroyo V, et al: Compensated cirrhosis: Natural history and prognostic factors. Hepatology. 1987, 7: 122-128.
Ginès P, Titó L, Arroyo V, et al: Randomized study of therapeutic paracentesis with and without intravenous albumin in cirrhosis. Gastroenterology. 1988, 94: 1493-1502.
Moore KP, Wong F, Ginès P, et al: The management of ascites in cirrhosis: Report on the consensus conference of the International Ascites Club. Hepatology. 2003, 38: 258-266.
Runyon BA. AASLD practice guidelines. Management of adult patients with ascites due to cirrhosis: an update. Hepatology. 2009, 49: 2087-2107.
Runyon BA, Montano AA, Akriviadis EA, et al: The serum-ascites albumin gradient is superior to the exudate-transudate concept in the differential diagnosis of ascites. Ann Intern Med. 1992, 117: 215-220.
Sanyal AJ, Genning C, Reddy KR, et al: The North American Study for the Treatment of Refractory Ascites. Gastroenterology. 2003, 124: 634-641.
Hepatic Encephalopathy
Blei AT, Cordobá J. Practice Parameters Committee of the American College of Gastroenterology: Hepatic encephalopathy practice guidelines. Am J Gastroenterol. 2001, 96: 1968-1976.
Bajaj JS, Saeian K, Schubert CM, et al: Minimal hepatic encephalopathy is associated with motor vehicle crashes. Hepatology. 2009, 50: 1175-1183.
Bustamante J, Rimola A, Ventura PJ, et al: Prognostic significance of hepatic encephalopathy in patients with cirrhosis. J Hepatol. 1999, 30: 890-895.
Cordobá J, Blei AT. Treatment of hepatic encephalopathy. Am J Gastroenterol. 1997, 92: 1429-1439.
Fitz JG. Hepatic encephalopathy, hepatopulmonary syndromes, hepatorenal syndrome, coagulopathy and endocrine complications of liver disease. In: Feldman M, Friedman LS, Sleisenger MH (eds): Sleisenger & Fordtran’s Gastrointestinal and Liver Disease. 8th ed Philadelphia: WB Saunders, 2006, pp 1966-1972.
Stahl J. Studies of the blood ammonia in liver disease: Its diagnostic, prognostic, and therapeutic significance. Ann Intern Med. 1963, 58: 1-24.
Variceal Hemorrhage
Garcia-Tsai G, Sanyal AJ, Grace N, Carey WD. Practice Guidelines Committee of American Association for Study of Liver Diseases; Practice Parameters Committee of the American College of Gastroenterology: Prevention and management of gastroesophageal varices and variceal hemorrhage in cirrhosis. Hepatology. 2007, 46: (3): 922-938.
Gotzsche PC, Hrobjartsson A: Somatostatin analogues for acute bleeding oesophageal varices. Cochane Database Syst Rev 2005, CD000193.
Groszmann RJ, Garcia-Tsao G, Bosch J, Grace ND, Burroughs AK, Planas R, et al: for the Portal Hypertension Collaborative Group. Beta-blockers to prevent gastroesophageal varices in patiens with cirrhosis. N Engl J Med. 2005, 353: 2254-2261.
North Italian Endoscopic Club for the Study and Treatment of Esophageal Varices: Prediction of the first variceal hemorrhage in patients with cirrhosis of the liver and esophageal varices. A prospective multicenter study. N Engl J Med. 1988, 319: 983-989.
Sharara AI, Rockey DC. Gastroesophageal variceal hemorrhage. N Engl J Med. 2001, 345: (9): 669-681.
Soares-Weiser K, Brezis M, Tur-Kaspa R, Leibovici L: Antibiotic prophylaxis for cirrhotic patients with gastrointestinal bleeding. Cochrane Database Syst Rev. 2002;(2):CD002907.